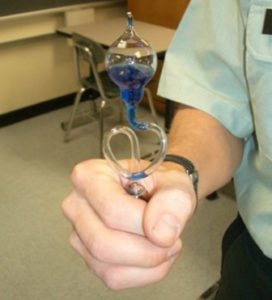
Model Description
This is a demonstration of how pressure increases with temperature particularly at temperatures above the fluid’s boiling point. Warming the bottom chamber beyond the boiling point increases the pressure sufficiently to displace fluid into the upper chamber. This demonstration should take 5 minutes.
Engineering Principle
The pressure of a fluid increases as you increase temperature. This relation is more dramatic when heating a vapor. For an ideal gas, the equation of state is governed by the equation below where P is the absolute pressure, v is the specific volume, R is the gas constant and T is the absolute temperature.
Warming the fluid within the hand boiler above its boiling point causes the pressure within the lower chamber to increase drastically, and this pressure is sufficient to displace the remainder of the fluid to the upper chamber.
What You Need
| Item | Quantity | Description/Clarification |
|---|---|---|
| Hand Boiler | 1 | Available online. |
How It’s Done
In Class: Grasp the hand boiler (shown above). The temperature in the lower chamber will rise to above the boiling point for the fluid very quickly, and this will cause the displacement of the fluid from the lower to the upper chamber. As you continue to grasp the hand boiler, vapor bubbles will rise through the glass straw giving the appearance that the upper chamber is boiling.
Observations: Students should be able to visualize the effect of raising the fluids temperature above the boiling point and the ensuing increase in pressure in the lower chamber.
Additional Application: Ask students how to get the fluid to return to the lower chamber without turning it over (you can hold the upper chamber).
See which students have the warmest hands by passing the boiler around and clocking the time required to move all the fluid to the upper chamber.